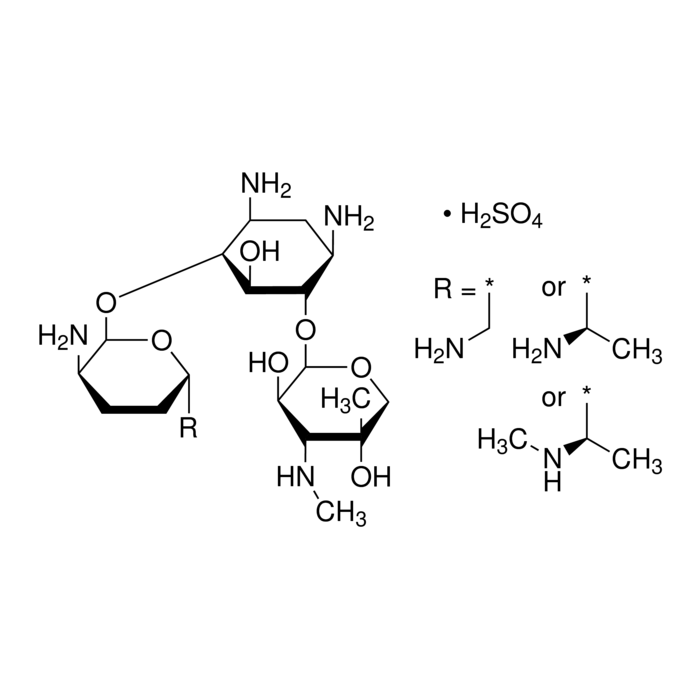
About Gentamicin Sulfate USP Grade, Gm
Gentamycin sulpahte is used as a selection agent (gentamicin-resistance gene) in molecular biology applications.It is used to treat many types of bacterial infections, particularly those caused by Gram-negative organisms.
Superior Quality and ComplianceGentamicin Sulfate USP Grade is carefully formulated to meet all relevant USP requirements, from purity to solubility. This guarantees consistency, safety, and performance in pharmaceutical manufacturing and laboratory research. Each parameter is stringently tested-from heavy metals (not more than 20 ppm) to endotoxin levels-to ensure a product of uncompromising quality.
Manufacturing in India for Global StandardsProduced in India, Gentamicin Sulfate USP Grade is offered by trusted manufacturers, suppliers, and traders. India's robust pharmaceutical sector ensures that each batch maintains international standards, making the product suitable for global distribution. Customers can rely on prompt supply and regulatory support when sourcing from Indian producers.
FAQ's of Gentamicin Sulfate USP Grade, Gm:
Q: How is the water content controlled in Gentamicin Sulfate USP Grade?
A: The water content in Gentamicin Sulfate USP Grade is precisely monitored to ensure it does not exceed 10%. This is achieved through stringent quality checks conducted during and after production, preserving the compound's stability and shelf-life.
Q: What is the assay method used for this product?
A: The assay of Gentamicin Sulfate USP Grade is performed using High Performance Liquid Chromatography (HPLC), a highly accurate method that ensures the precise quantification and purity assessment in every batch.
Q: When should Gentamicin Sulfate USP Grade be used in research or production?
A: This product is ideal for use when a high-degree of purity and compliance with USP standards is essential. It is commonly employed in pharmaceutical formulation and microbiological research where consistent efficacy and safety are required.
Q: Where is Gentamicin Sulfate USP Grade manufactured and supplied from?
A: Gentamicin Sulfate USP Grade is manufactured, supplied, and traded by reputable companies in India. The Indian pharmaceutical industry's adherence to strict global requirements ensures reliable product quality.
Q: What processes ensure the product meets USP and safety specifications?
A: Each batch goes through comprehensive testing, including identification, solubility, endotoxin levels, heavy metals, chloride content, and loss on drying, all in alignment with USP protocols. Advanced equipment and disciplined procedures safeguard product compliance.
Q: How can Gentamicin Sulfate USP Grade benefit your application?
A: Using USP Grade Gentamicin Sulfate secures high standards of purity, safety, and effectiveness. This is crucial for pharmaceutical manufacturing and laboratory research, where dependable performance is vital for reproducible results.