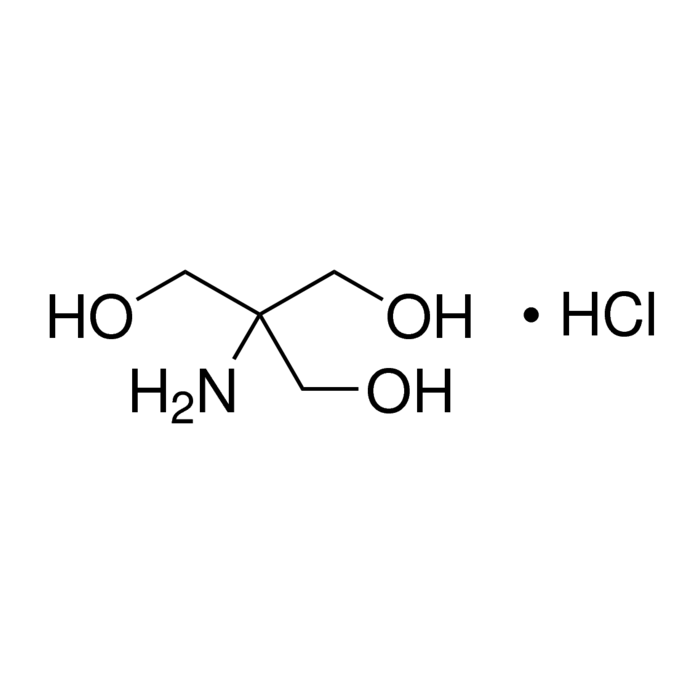
About Tris Hydrochloride, Reagent Grade, 99%
Tris and Tris Hydrochloride have been useful as buffers in a wide variety of biological systems. Uses include pH control in vitro and in vivo for body fluids and in buffering systems for electrophoresis applications. Tris has been used as a starting material for polymers, oxazolones (with carboxylic acids) and oxazolidines (with aldehydes). It can be used for the direct standardization of a strong acid solution; the equivalence point can be determined either potentiometrically or by use of a suitable indicator such as 3-(4-Dimethylamino-1-naphthylazo)-4-methoxybenzenesulfonic acid .
High-Purity Reagent for Precise ResultsTris Hydrochloride, Reagent Grade, 99%, offers exceptional purity, minimizing contamination in experimental results. Its analytical reagent standard guarantees suitability for critical research, including biochemistry, molecular biology, and analytical chemistry. As a non-hazardous, white powder, Tris-HCl is both safe to handle and convenient to use in laboratory workflows.
Reliable Buffer Preparation and SolubilityThis product dissolves readily in water, ensuring quick and complete solubilization for buffer and solution preparation. The consistent pKa of 8.1 (Tris base) allows precise pH control, which is crucial for enzyme reactions and other sensitive biochemical processes. Tris-HCl remains a staple in labs due to its reliable performance and ease of use.
FAQ's of Tris Hydrochloride, Reagent Grade, 99%:
Q: How should Tris Hydrochloride, Reagent Grade, 99% be used in buffer preparation?
A: Dissolve the appropriate quantity of Tris-HCl powder in water, adjusting the pH as needed. This process benefits from its excellent solubility, allowing for straightforward and reproducible buffer solutions commonly used in molecular biology and biochemistry.
Q: What are the main benefits of using Tris-HCl as an analytical reagent?
A: Tris-HCl, with its high purity and analytical reagent grade, provides reliable, reproducible results in laboratory applications. It is especially valued for its consistent pKa, water solubility, and non-hazardous nature, making handling and transportation safe and easy.
Q: When is Tris-HCl preferred over other buffering agents?
A: Tris-HCl is chosen when a stable buffer with a near-neutral pH (around 7-9) is required. Its pKa of 8.1 makes it ideal for many enzyme and molecular biology protocols where precise pH control is necessary.
Q: Where is Tris Hydrochloride, Reagent Grade, 99% sourced or manufactured?
A: This product is manufactured, supplied, and traded by reputable companies throughout India, ensuring reliable availability and quality compliance for research institutions and analytical laboratories.
Q: What is the process for dissolving Tris-HCl in water?
A: Simply measure the required amount of the white powder and add it to distilled water. Stir until completely dissolved. Adjust the pH if necessary, creating a buffer tailored to specific laboratory needs.
Q: How does the water solubility of Tris-HCl enhance laboratory applications?
A: Its rapid dissolution in water enables efficient and hassle-free preparation of solutions, ensuring consistent results and minimizing preparation time in a busy laboratory environment.